At present, NON-INVASIVE PRENATAL TESTING is referred to as NIPT, which is already well known by ordinary consumers on the land of China. NIPT has experienced three stages of non-regulation, suspension, and supervision. Starting from November 2016, all medical institutions and medical laboratories with relevant qualifications can carry out non-invasive DNA prenatal screening and diagnosis. National policies have been continuously optimized and guided by the industry. Order development. In order to improve population quality, eugenics and education, and promote the development of medical and health care, local governments not only proposed NIPT free programs (such as Changsha, Shenzhen, etc.), but also clarified their testing fee standards to make them open and transparent, even in many provinces and cities. All are actively working to include the NIPT project in medical insurance, thereby reducing the cost of consumers. With the dividends brought by the policy and the liberalization of the second-child policy, it is expected that the NIPT market will maintain a certain growth rate.
The rigid demand of NIPT continues to rise, the sample size continues to increase, and the timeliness required by consumers has put some pressure on prenatal screening centers, prenatal diagnosis centers, and third-party inspections. However, AMTK launched a series of liquid handling workstations. It serves the needs of NIPT high-throughput testing, and provides a complete set of mature and standard automation solutions for the NIPT project.
For NIPT testing with different throughputs and different kits, AMTK has launched a variety of automated solutions to fully match the NIPT needs of different customers.
NON-INVASIVE PRENATAL TESTING(Next Generation Sequencing Flow Chart)
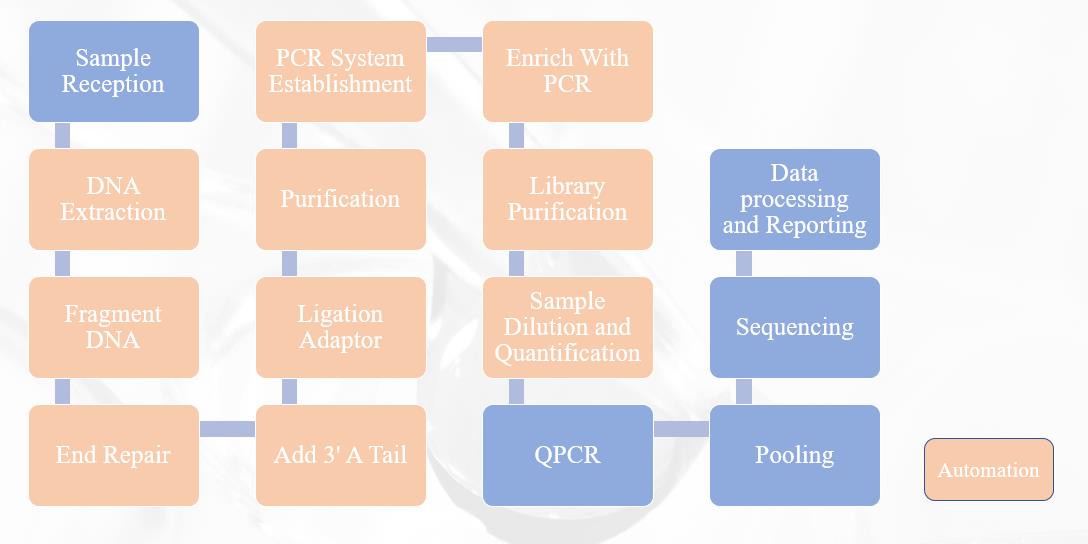
Workstation solution configuration
Instrument configuration
High-throughput solution configuration: This configuration can complete automated NIPT detection of 96-192 samples


Mid-throughput solution configuration: This configuration can complete automated NIPT detection of 32-96 samples

LH-1808
Low-throughput solution configuration: This configuration can complete automated NIPT detection of 1-32 samples
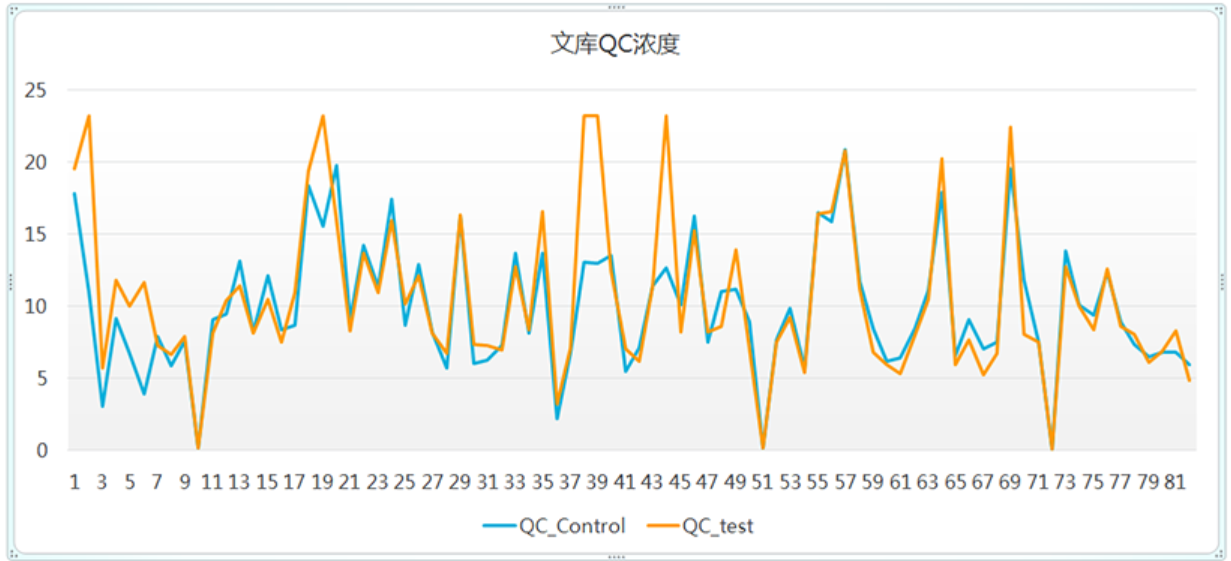
Experimental data case
After library construction, the concentration of 83 samples was manually compared with AMTK automated workstation data
Library QC concentration
Summary:
The data shows that there is no significant difference in QC concentration between manual (QC_CONTROL) and AMTK workstation (QC_TEST)